Introduction to Food Microbiology and Safety
The Essential Role of Cleaning Agents in Food Safety: Tackling Grime and Germs in Food Factories
In the heart of every food factory lies a relentless battle against grime and germs. To ensure pristine conditions and uphold food safety standards, cleaning and disinfecting agents are the unsung heroes, working tirelessly to maintain a safe and hygienic environment. Let’s delve into their crucial role in safeguarding food quality.
Let’s explore the fascinating world of cleaning agents and disinfectants in food factories. Imagine a microscopic battlefield, where grime and germs are the relentless adversaries. Our heroes? The cleaning and disinfecting agents, each equipped with unique powers to ensure cleanliness and food safety. Let’s see how they work.
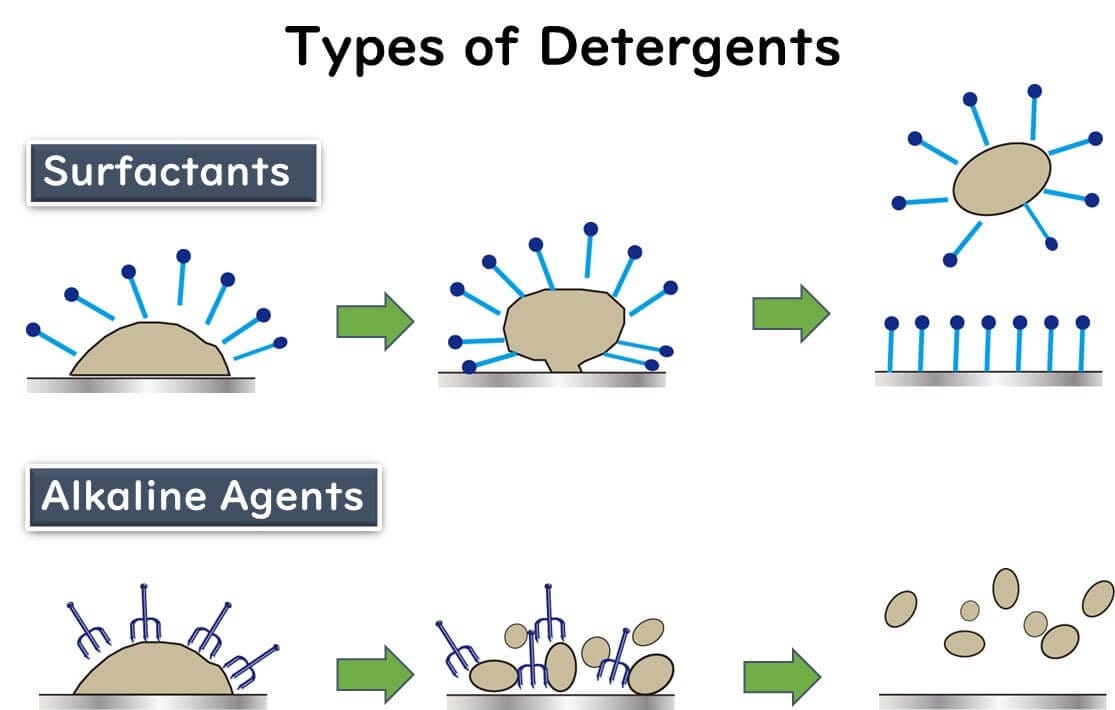
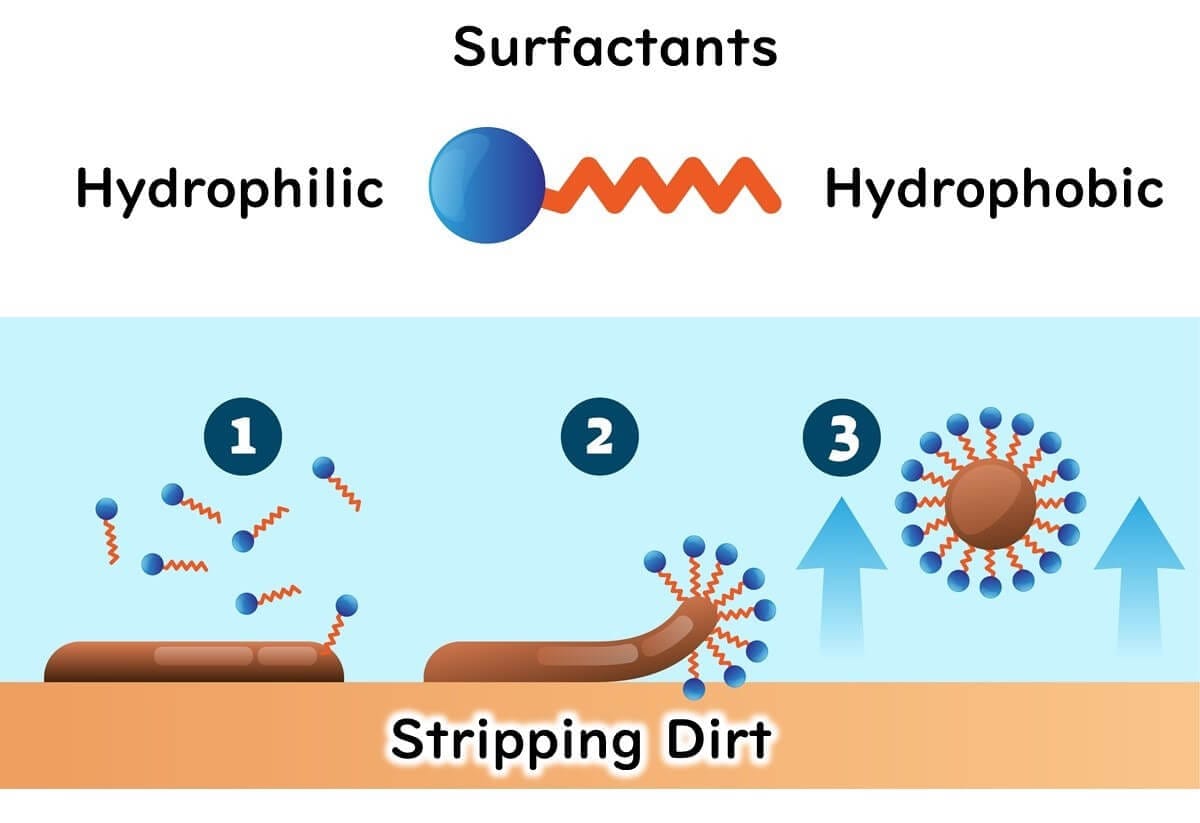
Surfactants: The Key to Tackling Oily Residues
Surfactants are like the double agents in spy movies. With their hydrophilic (water-loving) head and hydrophobic (oil-loving) tail, these molecules excel at breaking through oily residues commonly found in food processing environments. They work by surrounding dirt and grease on surfaces, lifting them away like a helicopter rescuing stranded debris. This action is critical for cleaning machinery and workstations effectively, setting the stage for proper disinfection.
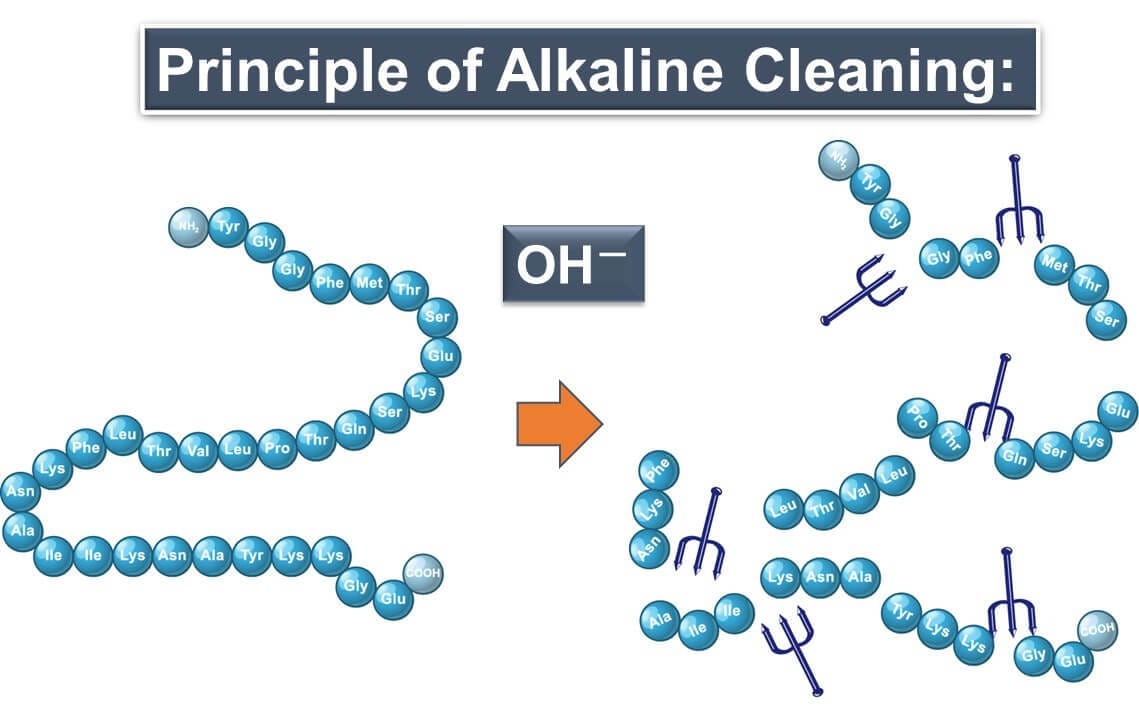
Alkaline Cleaners: Breaking Down Stubborn Contaminants
Alkaline cleaners are the powerhouses of cleaning agents. Packed with OH- ions, they don't just remove dirt; they break it down at a molecular level. These ions attack proteins and other organic residues, reducing them to smaller peptides or fragments. This process is essential for removing protein-based contaminants like meat juices or dairy residues, ensuring a thorough clean and a safe environment for food processing.
Related posts:
Why 70% Ethanol is the Star of Sterilization: Mechanisms and Limitations In the world of ethanol sterilization, 70% concentration is the...
Optimizing Sodium Hypochlorite Use in Food Factories: Key Insights and Practical Tips Explore the essential role of sodium hypochlorite in maintaining cleanliness...
Unlocking the Power of Acidic Electrolyzed Water in Food Safety Discover the science and applications of acidic electrolyzed water (hypochlorous...
The Role of Peroxyacetic Acid Formulations in Food Factory Disinfection Discover how peroxyacetic acid formulations stand out as robust disinfectants...
Categories

Discover expert-led lessons in food microbiology designed for professionals and beginners alike.

Author of this Blog: Bon Kimura
Bon Kimura, Professor Emeritus at Tokyo University of Marine Science and Technology (TUMSAT), specializes in food microbiology. He obtained his PhD from Kyoto University and became a professor at TUMSAT in 2006, serving as Dean of the Faculty of Marine Science from 2012-2015. Kimura has published over 200 international papers on food safety, pathogens, and spoilage bacteria. He has received multiple awards, including the Japanese Society for Food Microbiology Award (2019). Kimura also served as an editor for the International Journal of Food Microbiology from 2012 to 2024, where he was the principal reviewer for 1,927 papers.

Copyright © Introduction to Food Microbiology and Safety All Rights Reserved.